Probleemstelling:
According to current evidence, localized kidney tumors are best treated by partial nephrectomy to preserve as much functional renal tissue as possible without sacrificing survival chances. In many expertise centers, a robotic-assisted technique is currently preferred over open surgery, mainly due to its benefits regarding post-operative recovery.
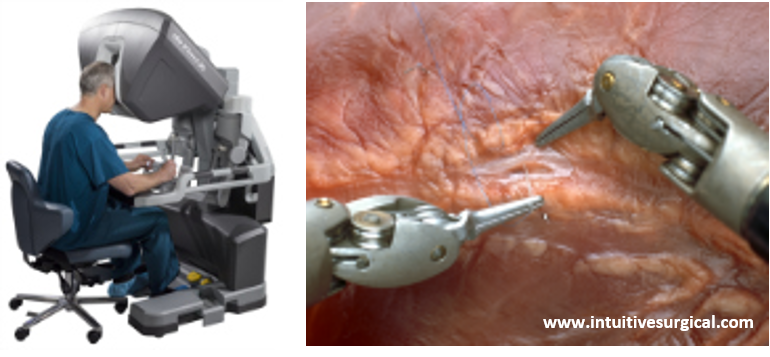
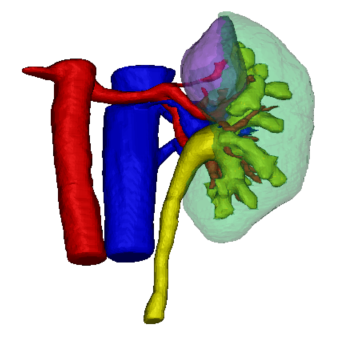
Currently, the robot surgery technique (Fig. 1) heavily depends on the limited intraoperative view and the 3D insight of the surgeon based on pre-operative imaging. Hence, 3D reconstructions could be really useful to assist intra-operatively in robot-assisted partial nephrectomy. 3D models could also be used to pre-operatively plan the surgical procedure. More specifically, the possible cutting locations should be investigated based on a detailed morphological analysis, while at the same time making sure that sufficient kidney function is preserved. Hence, the kidney should be still viable and foreseen of sufficient blood supply after partial nephrectomy.
The development of a planning tool that can help to determine the optimal surgical strategy (pre-operatively and intra-operatively) would thus be most welcome. In this way, chances for successful partial nephrectomies with negative cutting edges and maximum maintenance of healthy renal tissue would be higher. The starting point for such a planning tool is to obtain qualitative segmentations and 3D reconstructions of the patient-specific kidney structures of interest (being the parenchym, tumor, blood vessels etc.). Since manual segmentations are very time-consuming, this thesis will focus on automated segmentation methods.
Fig. 1. Illustrations of robot assisted surgery