Doelstelling:
The student will help with the development of an advanced flow bioreactor that can mimic the physiological shear stress and biaxial (circumferential and axial) stretch during physiological pulsatile flow. The separate components of the flow bioreactor (Pump, flowchamber, tubing and the macrovasculature design – see fig. A) will be adapted towards these physiological conditions based on computational fluid dynamics. For the adaptation of the macrovasculature design, the student will help with the modelling and the 3D bioprinting of the vessels in sterile conditions using the Regemat BioV1 3D bioprinter (see fig. B). Additionally, the student will optimize the sterile operation of the flow bioreactor: the system needs to be easy to assemble and it should be practical to refill the culture medium reservoir within a laminar flow cabinet. The different components of the flow system should be able to resist steam sterilization to guarantee sterility during the experiment. Also, the student will investigate the most optimal position within a CO2 incubator without disrupting the incubator operation. If necessary, an alternative for the CO2 incubator system needs to be found.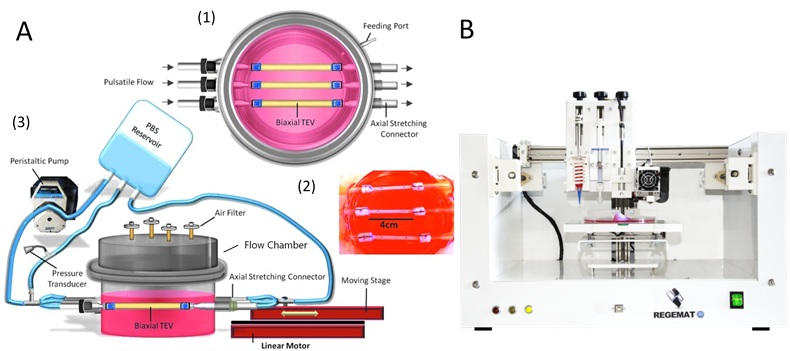
Figure 1: A) Illustration of a biaxial flow bioreactor system: (1) schematic top view of the flow chamber. (2) image of macrovasculature grafts inside the flow chamber. (3) A schematic of the flow chamber connected to both a linear motor and peristaltic pump to generate cyclic biaxial stretching. Adapter from huang et al (2016). B) Regemat BioV1 3D bioprinter